LYMPHOCYTE CLASSIFICATION
Lymphoma is a cancer of the lymphocyte
There are three types of lymphocyte — T cells, B cells, and natural killer (NK) cells — and any of these can give rise to a lymphoma
There are different techniques that can be used to identify specific lymphocytes and identify cancerous cells
H&E:
Standard H&E staining is the first line and can identify both atypical cells and abnormal patterns of lymphocyte infiltration in the skin.
In CTCL (particularly mycosis fungoides), the classic features are:
Atypical lymphocytes with hyperchromatic, irregular, cerebriform nuclei (so called because the convoluted nuclear membrane resembles the surface of the brain). When these cells circulate in the blood in Sézary syndrome they are called Sézary cells.
Epidermotropism — the tendency of these atypical T cells to migrate up into the epidermis. This is a hallmark feature of MF and helps distinguish it from inflammatory mimics, where lymphocytes are usually confined to the dermis. Importantly, the epidermis itself is typically not spongiotic, despite being infiltrated — a useful clue.
Pautrier microabscesses — small intraepidermal collections of these atypical lymphocytes. They are highly specific for MF but only seen in a minority of cases (around 10–20%), so their absence does not exclude the diagnosis.
Immunohistchemistry:
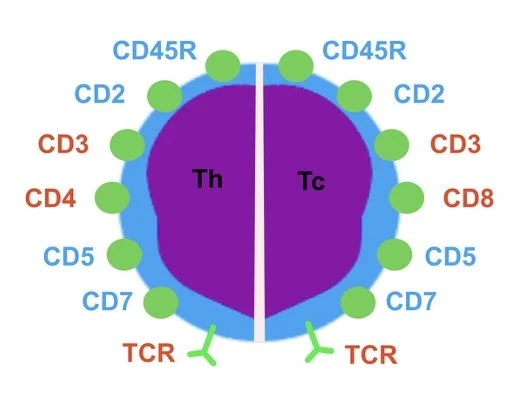
We identify different lymphocytes by the CD markers (proteins) on their surface — think of these as flags. They can be identified by using immunohistochemical stains (antibodies which attach to the marker and stain brown)
The presence or absence of certain proteins helps us decide what types of cells are present and if they are normal or abnormal
For instance:
CD3 marks T cells
CD20 marks B cells
T cells can be subdivided into CD4+ helper cells, CD8+ cytotoxic cells, and regulatory T cells
T cells also normally express a set of pan-T cell markers:
CD2
CD3
CD5
CD7
— meaning these are present on most mature T cells. However, when a T cell becomes malignant, it can lose expression of one or more of these markers.
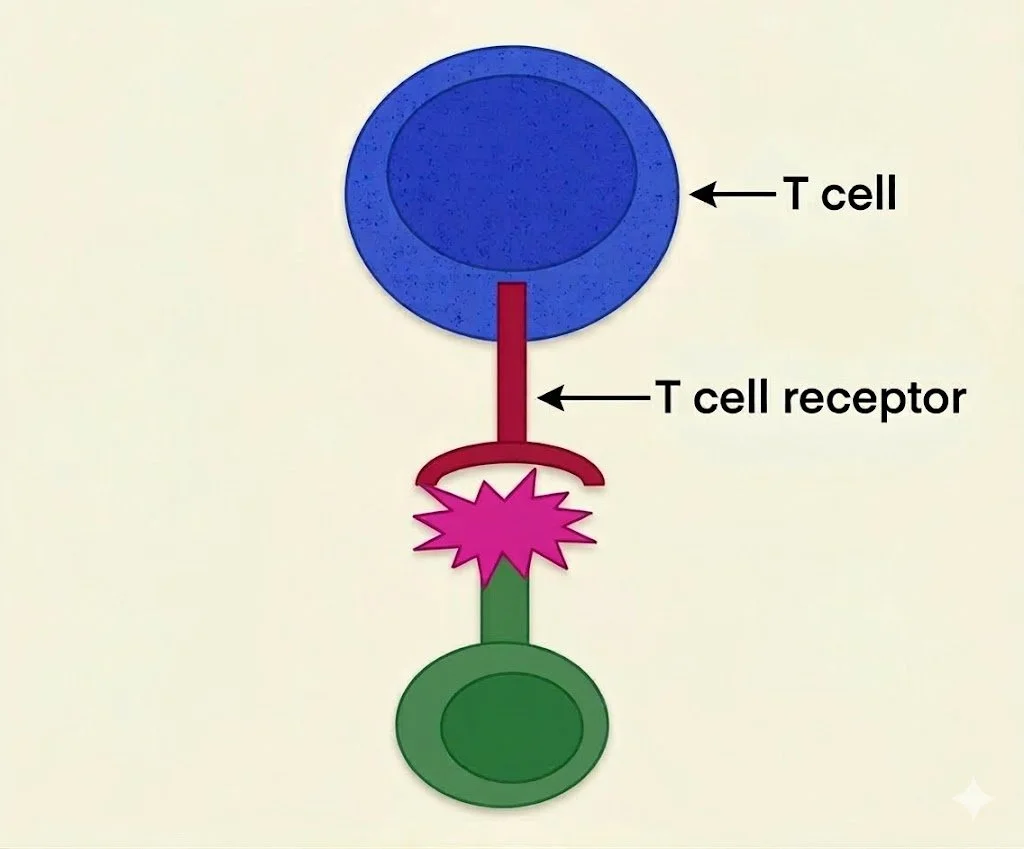
Surface receptors and clonality
Lymphocytes also carry unique surface receptors
T cells have a T cell receptor (TCR) — this is the part of the T cell that binds to antigens and activates the immune system.
Every T cell has a slightly different receptor, which is how the immune system can recognise the huge variety of antigens it encounters.
In normal tissue you see a diverse mix of these receptors.
When one cell multiplies out of control, you find clones — many cells with identical receptors
T cell receptor gene rearrangement
This is the test we do to look for a clone (clonality)
You may see this test referred to by different names — TCR gene rearrangement, high-throughput sequencing, or PCR — but they are all doing the same thing: looking to see if one T cell clone is multiplying out of control
TCR gene rearrangement studies can be performed by using PCR on DNA from lymphocytes extracted from various tissues (eg skin, blood, bone marrow)
In normal tissue, T cells all have slightly different receptors, so when you run the test you see a smooth waveform with a normal distribution — indicating lots of different T cells with no single dominant clone
If one cell starts to multiply, you'll see a small peak emerging from the waveform. As that clone continues to expand, the peak becomes larger and more dominant, while the diversity of the other lymphocytes drops away. This is reported as a monoclonal peak and supports a diagnosis of lymphoma
It is important to remember that all cancers are clones, but not all clones are cancer
A monoclonal peak can also be seen in benign reactive processes (such as infections, drug reactions, or eczema), where you would expect the clone to disappear once the trigger resolves
Equally, a clone can sometimes be missed — if the disease is very early or the sample is too small or degraded
B cell receptor (BCR) studies
B cells also have a B cell receptor (BCR) — which is essentially the membrane-bound form of the immunoglobulin (antibody) that the B cell would secrete
There are two main ways we look for a B cell clone:
Immunoglobulin gene rearrangement (IGH and IGK) — the B cell equivalent of TCR gene rearrangement. PCR is used to amplify the rearranged immunoglobulin heavy chain (IGH) and kappa light chain (IGK) genes, and a monoclonal peak suggests a B cell lymphoproliferative process. Testing both IGH and IGK together pushes the detection rate close to 99%
It can be useful to test both as certain lymphomas (eg follicular centre–derived lymphomas) can give false negatives if IGH is tested alone.
Light chain restriction — a simpler, complementary test usually done by immunohistochemistry or flow cytometry. Every B cell makes antibodies with either a kappa or a lambda light chain (never both). In a normal reactive infiltrate you see a mix of kappa- and lambda-expressing B cells in a roughly 2:1 ratio. If virtually all the B cells are expressing the same light chain (i.e. the ratio is heavily skewed — "kappa-restricted" or "lambda-restricted"), that tells you they are all descended from a single parent cell and supports a clonal B cell process.
Flow cytometry
Is a way of counting and sorting lymphocytes based on the CD markers they express on their surface. In CTCL it is mainly used on blood to look for circulating malignant cells, though it can also be performed on other tissues such as lymph node or bone marrow.
The sample is put into suspension and individual cells are passed through a laser one at a time. Each cell is tagged with multiple fluorescent antibodies of different colours that target different CD markers, so multiple markers can be detected simultaneously on each cell in a single run — you get CD4 counts, CD8 counts, CD4:CD8 ratio, and marker loss information all at once.
This is useful because normal T cells express a predictable set of surface proteins including CD7 and CD26, and malignant T cells often lose one or more of these. Flow cytometry detects abnormal cells by identifying what's missing.
Key findings in CTCL with blood involvement (ie Sézary syndrome or erythrodermic MF) include an increased CD4:CD8 ratio (≥10), loss of CD7 and/or CD26 on CD4+ cells, and an elevated Sézary cell count
Putting it all together
Going forward, when working up a suspected cutaneous lymphoma, we rarely rely on a single test
The diagnosis is built up from a combination of:
H&E — to look at the morphology of the cells and the pattern of infiltration (eg epidermotropism, cerebriform nuclei, Pautrier microabscesses)
Immunohistochemistry — to phenotype the infiltrate and look for loss of normal T cell markers
TCR gene rearrangement studies — to demonstrate clonality
Flow cytometry — added in when we are thinking about blood involvement (eg suspected Sézary syndrome or erythrodermic MF)
No single one of these is diagnostic on its own — clones can occur in reactive conditions, marker loss can be partial, and morphology can be subtle, especially in early disease
It is the clinicopathological correlation of all the pieces together — alongside the clinical picture — that gets you to the diagnosis
CUTANEOUS LYMPHOMA CLASSIFICATION
Can divide all types of lymphoma in to Hodgkin’s and Non Hodgkin’s lymphoma
Systemic lymphomas:
The majority of systemic non-Hogdkin’s lymphomas tend to be B cell lymphomas whereas the majority of primary cutaneous lymphomas are T cell lymphoma
If have a lymphoma on the skin you want to think:
Is this a primary cutaneous lymphoma?
or
Is it a systemic lymphoma that has spread to the skin (secondary cutaneous lymphoma)?
Primary cutaneous T cell lymphomas
Primary cutaneous T cell lymphomas account for ≈ 80% of primary cutaneous lymphomas
BPDCN - blastic plasmacytoid dencritic cell neoplasm
As discussed in the introduction, we can identify a T cell lymphoma by the presence of CD3 on immunohistochemistry — this is the key pan-T cell marker.
Once you've confirmed you're dealing with a T cell process, primary cutaneous T cell lymphomas can be subdivided into three main groups
'Classic' CTCL
This is the largest group and includes mycosis fungoides (the commonest cutaneous lymphoma overall, accounting for ~50% of all primary cutaneous lymphomas), its variants (folliculotrophic MF, pagetoid reticulosis, granulomatous slack skin), and Sézary syndrome (~3%)
CD30+ lymphoproliferative disorders (~20%)
Defined by the expression of CD30 on the cell surface
This subgroup is staged and treated differently to MF and includes primary cutaneous anaplastic large cell lymphoma (pc-ALCL) and lymphomatoid papulosis (LyP)
Rare subtypes (~7%) — a collection of less common entities including:
CD8+ aggressive epidermotropic T cell lymphoma
NK-T cell lymphoma, nasal type
Gamma/delta T cell lymphoma
CD4+ small/medium T cell lymphoproliferative disorder (renamed)
Subcutaneous panniculitis-like T cell lymphoma
Adult T cell leukaemia/lymphoma
Acral CD8+ T cell lymphoma (provisional entity)
Primary cutaneous peripheral T cell lymphoma, NOS
This distinction matters clinically because each group has a different prognosis and management approach. Classic CTCL and CD30+ LPD tend to behave more indolently, whereas several of the rare subtypes can be aggressive
Primary cutaneous B cell lymphoma
Primary cutaneous B cell lymphomas account for ≈ 20% of primary cutaneous lymphomas
We can identify a B cell lymphoma by the presence of CD20 on immunohistochemistry
Blastic plasmacytoid dendritic cell neoplasm (BPDCN)
BPDCN is a rare and aggressive neoplasm derived from plasmacytoid dendritic cells — a completely different cell lineage to T and B lymphocytes
It is classified separately within the primary cutaneous lymphoma framework for this reason and will be discussed later in more detail
MYCOSIS FUNGOIDES
Is the most common form of CTCL and accounts for 50% of all primary cutaneous lymphomas
CLINICAL
Typically affects older adults (median age at diagnosis: 55-60 years)
Can occur in children (including a hypopigmented variant)
Usually follows an indolent course with slow progression over years or even decades
General progression: patch → plaque → tumour
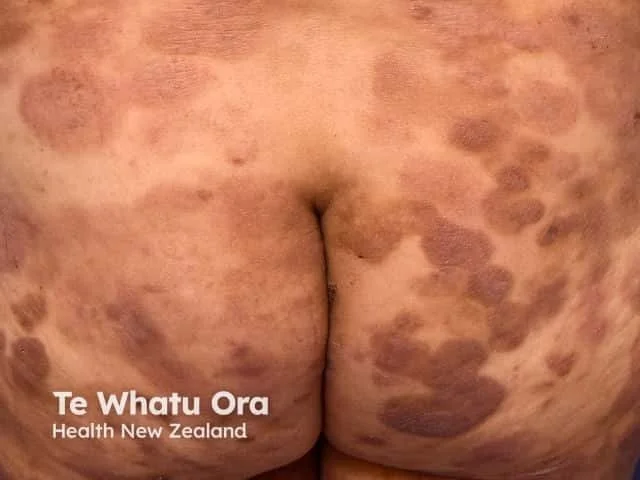
Initial lesions have a predilection for the buttocks and other sun-protected areas (“bathing suit distribution”)
In later stages, lymph nodes and visceral organs may become involved
Patch stage:
Flat, erythematous lesions with fine scale
Surface atrophy with fine wrinkling may be seen
Often asymmetrical
Irregular “geometric” shaped lesions
Sometimes hair loss over sites of disease
May improve in sun and show some (slow, partial) response to topical steroids
Often a long history of slow evolution — frequently misdiagnosed as eczema, psoriasis, or tinea
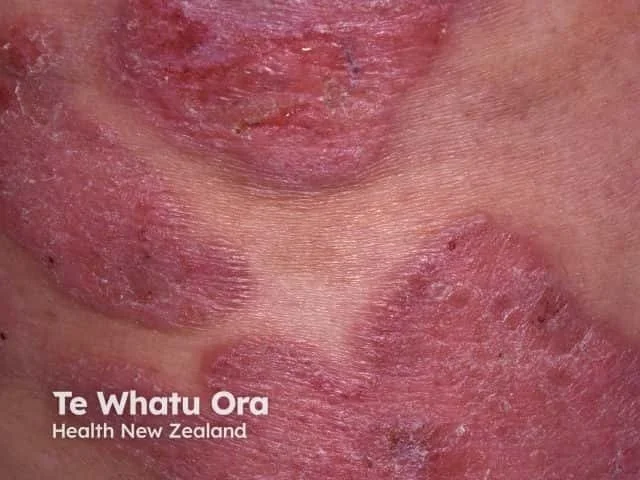
Plaque stage:
Raised, well-demarcated, often violaceous plaques
Thicker and more infiltrated than patches
May co-exist with patches
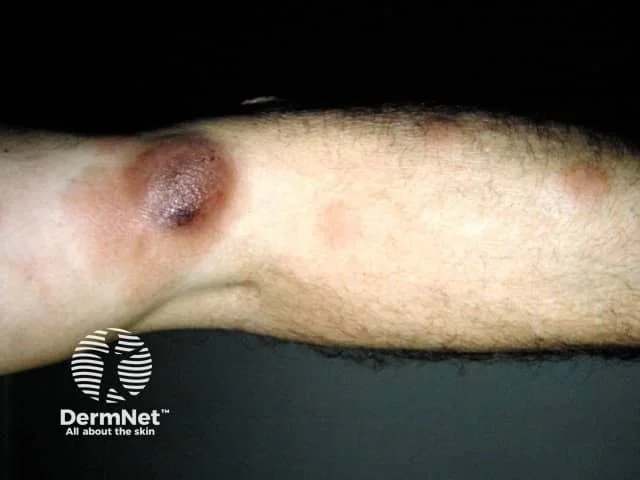
Tumour stage:
Dome-shaped nodules or tumours (≥1 cm)
May ulcerate
Can arise on a background of pre-existing patches and plaques — always look for coexisting patches/plaques
Transformation to a diffuse large cell lymphoma (CD30+ve or −ve) can occur and carries a poor prognosis
Distribution:
Pelvic girdle and shoulder girdle; sun-protected sites
Asymmetrical distribution is characteristic
HISTOPATHOLOGY
Early patch:
Superficial bandlike infiltrates consisting of lymphocytes and histiocytes
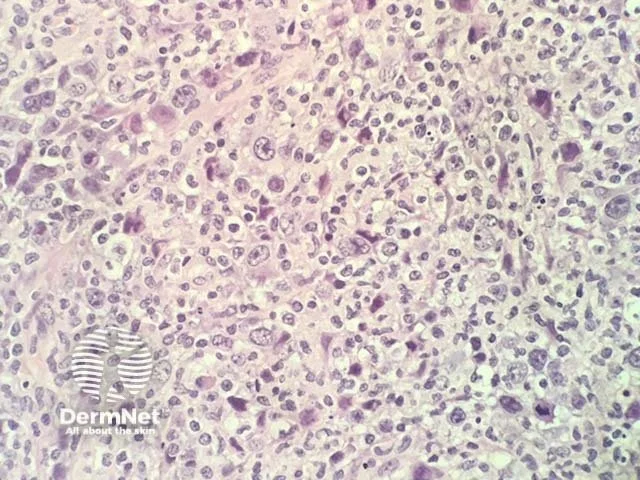
Can get atypical cells with small to medium sized, highly indented (cerebriform), hyperchromatic nuclei
These atypical cells are mostly confined to epidermis (epidermotropism) and may initial be seen around basal layer
Patch:
Superficial band-like infiltrate of lymphocytes and histiocytes are seen in the upper dermis
Atypical lymphocytes — a few small to medium-sized cells with cerebriform, hyperchromatic nuclei (convoluted, looks like a raisin) infiltrate into the epidermis (epidermotropism) and importantly the epidermis is not spongiotic despite being infiltrated (a useful clue to MF)
Classically, the atypical cells are intially found in the basal layer and appear single "haloed" cells (lymphocyte surrounded by small clear vacuole) which then can begin to ‘line up’ up along the dermo-epidermal junction
Pautrier’ microabscesses: Small clusters of atypical lymphocytes within the epidermis. These are pathognomonic but are actually less common in the patch stage than in the plaque stage.
Plaque:
As the disease progresses to a plaque, the cellular infiltrate becomes more "heavy" and the architectural distortion is more obvious.
The epidermotropism becomes more pronounced
May see more Pautrier microabscesses
A denser dermal infiltrate will be seen
More obvious epidermotropism and denser dermal infiltrate
Pautrier microabscesses
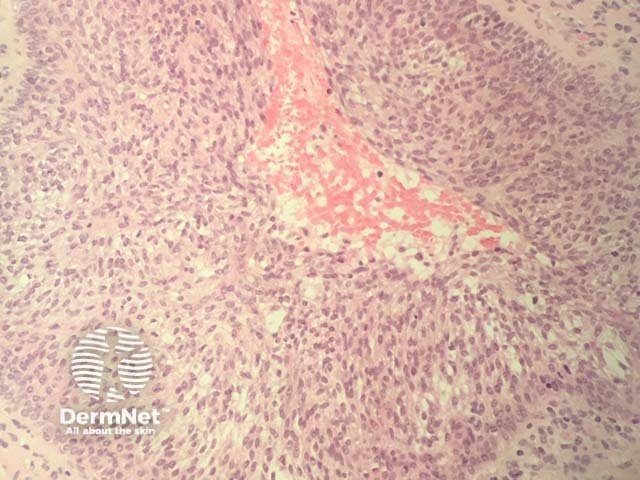
Tumour:
The dermal infiltrate becomes more diffuse and epidermotropism may be lost
The tumour cells increase in size and numbers
Cells can become more pleomorphic
Transformation to a diffuse large cell lymphoma (CD 30 +ve or -ve) can occur and has a poor prognosis
Dense dermal infiltrate in tumour stage
Large cell transformation - poor prognosis
IMMUNOHISTOCHEMISTRY
CD3+, CD4+, CD45RO +ve (CD8 -ve usually)
Rarely can be CD4- and CD8+ (particularly in hypopigmented disease)
Can lose pan-T cell antigens (CD2, CD3 and CD5) which can aid in diagnosis
CLONALITY
Clonal T-cell receptor gene arrangement is detected in most cases of MF
Tips regarding TCR gene rearrangement in MF:
The detection of a T cell clone in skin in the absence of consistent histology is NOT diagnostic of CTCL
The presence of an identical T cell clone in multiple skin biopsies (± blood) is useful in confirming a diagnosis of CTCL in patients with consistent histology and clinical features
In patients where diagnosis is uncertain, identification of identical clones from 2 separate skin biopsies helps to confirm CTCL
DNA from different biopsies and different tissues should always be analysed together to provide informative results
T cell clones in the blood alone (especially in older patients) can be incidental and are not necessarily diagnostic
MF VARIANTS
FOLLICULOTROPHIC MF
This is a variant of MF characterised by a folliculotrophic infiltrate, often with sparing of the epidermis. It frequently affects the head and neck. Most cases (but not all) show mucinous degeneration of the hair follicles (follicular mucinosis). Because infiltrates are associated with the follicles and therefore deeper, skin-directed therapies are often less effective.
Clinical:
Mostly seen in adults (can affect children)
Males > Females
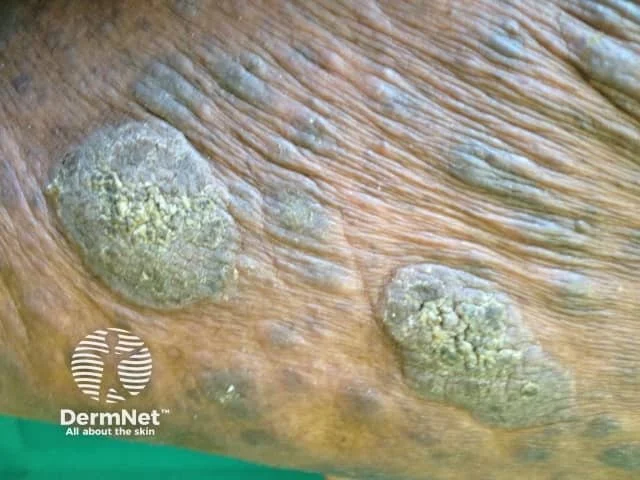
Can present as grouped follicular papules, acneiform lesions, or indurated plaques/tumours
Often associated with alopecia and release of clear mucin from affected areas (mucinorrhoea)
Can be very itchy — progressing pruritus may indicate disease progression
Histopathology:
Perivascular and periadnexal infiltrates with variable infiltration of follicular epithelium by small, medium, and sometimes large hyperchromatic cells with cerebriform nuclei
Epidermis tends to be spared
Alcian blue stains may demonstrate mucinous degeneration of follicular epithelium (“follicular mucinosis”)
May see eosinophils and plasma cells
If eccrine glands are prominently affected it is called syringotropic MF
Folliculotrophic MF
Immunohistochemistry:
Most cases CD3+, CD4+, CD8-
May also see CD30+ blast cells
Prognosis:
Disease specfic 5-year survival: 70-80%
Similar to that of classical tumour stage
Treatment issues:
As the infiltrates are associated with the follicles they are often deeper and as such skin directed therapies tend to be not as effective
PAGETOID RETICULOSIS
Pagetoid reticulosis is a rare, indolent variant of mycosis fungoides recognised in the WHO–EORTC classification of cutaneous lymphomas. It typically presents as a solitary, slowly progressive hyperkeratotic or psoriasiform plaque on a distal extremity, with striking epidermotropism on histology and an excellent prognosis.
The disease is defined by its localised form (Woringer–Kolopp); historically there was a classification of a disseminated Ketron–Goodman variant but this has now been reclassified under different aggressive CTCLs.
Clinical:
Solitary psoriasiform or hyperkeratotic patch or plaque, often localised to the extremities
Slowly progressive
Histopathology:
Hyperplastic epidermis with marked infiltration by atypical pagetoid cells (singly or in nests)
Medium or large sized hyperchromatic and cerebriform nuclei with abundant, vacuolated cytoplasm
Upper dermis may show mixed lymphoctes or histiocytes but no neoplastic T-cells
Immunohistochemistry:
CD3+, CD4+, CD8- (or CD4-, CD8+)
CD30 often expressed
Treatment:
Responds well to radiotherapy
Surgical excision can be considered
Topical nitrogen mustard or topical steroids can be used in some instances
Prognosis:
Extracutaneous dissemination or disease-related deaths not reported
GRANULOMATOUS SLACK SKIN
A very rare variant of MF characterised by the slow development of folds or lax skin in the major skin folds
Clinical:
Circumscribed areas of lax, pendulous skin with predilection for the axillae and groin
One-third of reported cases have an association with Hodgkin’s disease
Association with classical MF also reported
Most cases have an indolent course
Histopathology and immunohistochemistry:
Dense granulomatous dermal infiltrates containing atypical T cells with indented to cerebriform nuclei
Macrophages and multinucleated giant cells may be seen
Destruction of elastic tissue
Epidermis may show focal infiltration by small atypical T cells
CD3+, CD4+, CD8−
Treatment:
Radiotherapy may help but experience limited
Rapid recurrence after excision has been reported
SEZARY SYNDROME
Sézary syndrome is the leukaemic variant of CTCL defined by a classic triad:
Erythroderma
Generalised lymphadenopathy
Neoplastic T cells (Sezary cells) in skin, lymph nodes and peripheral blood
Clinical:
Rare; exclusive to adults
Erythroderma which may be associated with marked exfoliation, oedema, lichenification, and intense pruritus
Common associated findings:
Lymphadenopathy
Alopecia
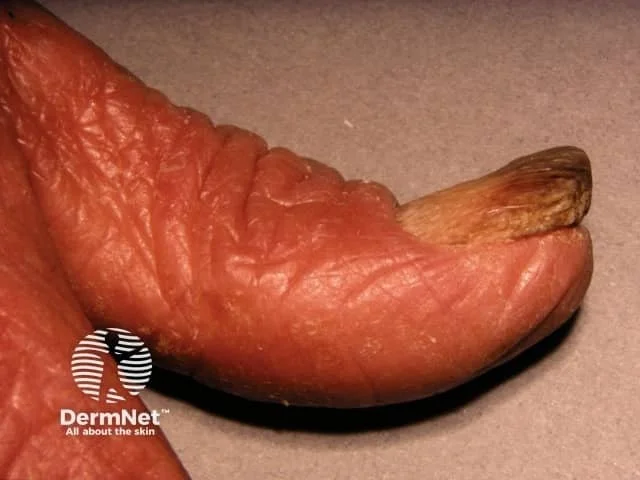
Onychodystrophy (nail dystrophy)
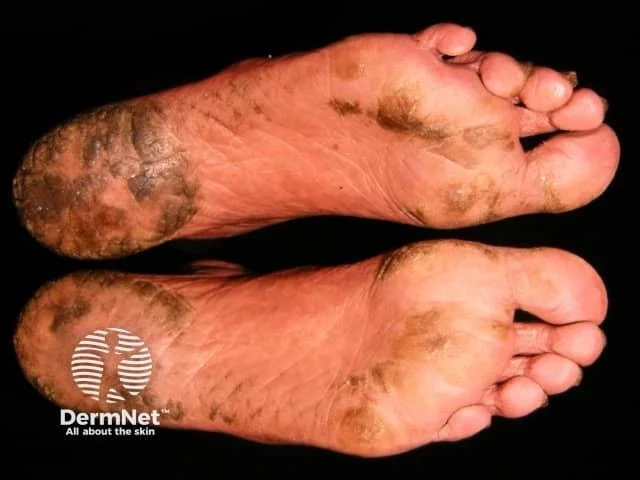
Palmoplantar hyperkeratosis
Clinical tip: The erythrodermic patient can be difficult to diagnose. Look for palmar/plantar keratoderma and nail dystrophy. Ask the patient about the skin before they developed the erythroderma and ask for old photographs — these may reveal preceding patches of MF which can help clinch the diagnosis.
Making the diagnosis:
Clinical criteria:
The “classic triad” must be present for a clinical suspicion of SS:
Erythroderma: Generalized redness (erythema) involving at least 80% of the total body surface area (BSA)
Lymphadenopathy: Enlargement of peripheral lymph nodes
Circulating Malignant Cells: Presence of atypical T-cells with "cerebriform" (brain-like) nuclei, known as Sezary cells
Haematologic (blood) criteria
To define B2 blood involvement and distinguish Sézary syndrome from erythrodermic mycosis fungoides, we need:
A clonal TCR rearrangement in the blood matching the skin clone
Plus
Evidence of a high tumour burden in the blood
The current EORTC/ISCL/USCLC consensus (2022) defines B2 by flow cytometry using absolute counts. B2 is met when there are:
≥1000/µL clonal CD4+ T cells identified by an aberrant phenotype — either CD4+CD7− or CD4+CD26− populations
Plus a positive blood T-cell clone matching the skin.
Previous (2007) ISCL definitions also accepted:
A CD4:CD8 ratio ≥10
or
Percentage-based aberrant phenotypes (≥40% CD4+CD7− or ≥30% CD4+CD26−) as alternative B2 criteria
These have been removed from the 2022 update because they can detect skewed CD populations rather than true tumour burden
Manual cerebriform Sézary cell counts on a blood film are also no longer preferred due to poor inter-observer reproducibility
The 2022 criteria are more specific for Sezary Syndrome but a recent study (Chrisman et al., Blood 2024) suggests they may miss a small subset of Sézary patients with low blood burden who nonetheless have a poor prognosis — so this is an active area of debate
Skin biopsy:
Findings overlap with mycosis fungoides: a band-like (lichenoid) infiltrate of small-to-medium atypical lymphocytes with cerebriform nuclei in the upper dermis, with epidermotropism and occasional Pautrier microabscesses.
However, compared to MF the infiltrate and epidermotropism tends to be much sparser and often the biopsies an be quite non-specific
Up to a third of biopsies are non-diagnostic, so multiple biopsies and clinicopathological correlation are often needed.
Immunohistochemistry typically shows a CD3+ CD4+ CD8− CD45RO+ memory T-cell phenotype with frequent loss of CD7 and CD26.
Lymph node biopsy:
Excisional biopsy of the largest palpable node is preferred over fine-needle aspiration. Demonstrating the same T-cell clone in node and skin supports a diagnosis of SS.
Bone marrow:
Not routinely required for diagnosis or staging in SS, as marrow involvement is uncommon and rarely changes management
Reserved for cases with unexplained cytopenias or suspected transformation/second haematological malignancy
BLOOD INVESTIGATIONS IN CTCL WORKUP
Standard panel (all suspected CTCL)
FBC with differential — High lymphocyte count can be early marker of blood involvement
Peripheral blood film — to look for cerebriform Sézary cells; a positive film prompts flow cytometry but a negative film does not exclude blood involvement
U&E, LFTs, calcium — baseline
LDH — elevated LDH is associated with advanced disease and worse prognosis
HTLV-1 — to exclude HTLV-1-associated adult T-cell leukaemia/lymphoma which can present similarly to MF/SS and is more aggressive
β2-microglobulin — additional prognostic marker, especially in advanced stages
If Sézary syndrome or advanced MF is suspected, add:
Flow cytometry to assess tumour burden in blood
TCR gene rearrangement to demonstrate clonality and allow comparison with the skin clone
Immunoglobulins — hypogammaglobulinaemia is common in SS and increases infection risk
MAKING A DIAGNOSIS
Making the diagnosis is the key first step in managing MF/SS, and it often takes time to secure.
CTCL is notorious for mimicking benign inflammatory dermatoses, and the diagnostic work usually starts with history and skin examination, paying close attention to clues that point in the right direction, for example:
Patches in sun-protected sites (the "bathing-trunk" distribution) evolving over years through patches → plaques → tumours (MF)
Or an erythrodermic patient with alopecia, nail dystrophy, ectropion, and marked lymphadenopathy (SS)
Repeat biopsies are often needed — early MF in particular is histologically subtle, and a single non-diagnostic biopsy does not exclude the disease.
Studies that help confirm CTCL on tissue include H&E histopathology, immunohistochemistry (T-cell markers and aberrant loss of CD7/CD26), and TCR gene rearrangement studies to demonstrate clonality.
Where appropriate — particularly with erythroderma, suspected SS, or advanced MF — the workup extends beyond the skin: peripheral blood film, flow cytometry, and blood TCR rearrangement to assess for blood involvement, and excisional biopsy (favoured over FNA) of any palpable lymph node to look for nodal disease and matching clonality.
Once the diagnosis is clinched, the next key step is staging, which determines prognosis and guides treatment.
STAGING
Once the diagnosis of MF/S is made, staging defines the extent of disease which helps define prognosis and guide treatment.
The system used worldwide is the ISCL/EORTC TNMB classification
First published by Olsen et al. in 2007 and updated in 2022
MF/SS is staged differently from all other cutaneous lymphomas
It assesses four compartments:
T — skin
N — lymph nodes
M — viscera
B — blood
The depth of staging investigations should match the disease, eg:
A patient with a few patches just needs skin exam and basic bloods
A patient with erythroderma or lymphadenopathy will need imaging, lymph node biopsy and flow cytometry.
Skin (T)
Skin involvement depends on lesion type (patch, plaque, tumour, erythroderma) and body surface area (BSA).
A useful rule of thumb at the bedside: the patient's palm plus all five fingers ≈ 1% BSA
MF / Sézary Syndrome — T Staging (ISCL/EORTC)
Skin involvement classification.
| T | Definition |
|---|---|
| T1 | Patches/plaques covering <10% BSA |
| T2 | Patches/plaques covering ≥10% BSA |
| T3 | One or more tumours (≥1 cm), regardless of BSA |
| T4 | Erythroderma (≥80% BSA) |
T1 and T2 can be sub-stratified into a (patch only) or b (plaque ± patch). This does not change stage but carries prognostic weight — patients with plaques have a worse outcome than those with patches alone
Clinical tip: A single nodule >1 cm in a patient with otherwise widespread plaques moves them from T2 to T3 and from early to advanced stage — a major prognostic and management shift
The modified Severity-Weighted Assessment Tool (mSWAT) quantifies skin tumour burden by weighting lesion type (patch ×1, plaque ×2, tumour ×4) against regional BSA. Score ranges 0–400, and it is the standard tool for monitoring response
Lymph Nodes:
Should assess for any lymph nodes clinically
Any peripheral node >1.5 cm, or firm/irregular/fixed/clustered, is considered clinically abnormal and warrants further evaluation
CT neck/chest/abdomen/pelvis (with contrast) is indicated for :
T3 (tumour-stage)
T4 (erythroderma)
Palpable peripheral lymphadenopathy
B-symptoms
Abnormal bloods (raised LDH, cytopenias)
It can also be consider in T2b disease (patches/plaques ≥ 10% BSA)
MF / Sézary Syndrome — N Staging (ISCL/EORTC)
Lymph node involvement classification.
| N | Definition |
|---|---|
| N0 | No clinically abnormal peripheral nodes |
| N1 | Clinically abnormal — pathology does not definitively show lymphoma (dermatopathic) |
| N2 | Early lymphomatous involvement, architecture preserved |
| N3 | Partial or complete effacement of nodal architecture |
| Nx | Abnormal but no histological confirmation |
Note: A "clinically abnormal" peripheral node is defined as ≥1.5 cm in long axis (or firm, irregular, fixed, or clustered on examination). On imaging, the corresponding threshold is ≥1 cm in short axis.
Clinical tip - FNA is inadequate:
Nodal architecture must be assessed for staging
Excisional or core biopsy is the gold standard, taken from the most clinically or PET-avid node
Both H&E and TCR clonality should be requested
Visceral involvement:
MF / Sézary Syndrome — M Staging (ISCL/EORTC)
Visceral involvement classification.
| M | Definition |
|---|---|
| M0 | No visceral involvement |
| M1 | Histologically confirmed visceral involvement |
Common sites for visceral spread are lung, liver, spleen and CNS.
Bone marrow involvement was formally defined within visceral evaluation in the 2022 ISCL update, and bone marrow biopsy is recommended where there is unexplained cytopaenia or where systemic involvement would change management.
Blood (B):
Blood classification is what distinguishes Sézary syndrome from erythrodermic MF, and its definition has been refined twice since 2007
The 2022 ISCL/USCLC/EORTC update (Olsen, Blood 2022) is the current standard
Flow cytometry has replaced morphological Sézary cell counting — manual counts are subjective and poorly reproducible.
Blood involvement is measured by counting the abnormal (clonal) Sézary cells circulating per microlitre. These cells are identified on flow cytometry by their loss of normal T-cell markers — typically CD4+ cells that have lost CD7 or CD26
MF / Sézary Syndrome — B Staging (2022 ISCL/USCLC/EORTC)
Blood involvement classification.
| B | 2022 ISCL/USCLC/EORTC definition |
|---|---|
| B0 | <250/μL CD4+CD7− or CD4+CD26− cells |
| B1 | 250 to <1000/μL |
| B2 | ≥1000/μL with a clonal blood TCR matching the skin clone |
Each B level can be further sub-classified a (no clone, or clone not matching skin) or b (clone matching skin)
B0b confers a worse prognosis than B0a in early-stage disease, even though both are technically B0
Pearl: B classification has limited prognostic relevance in patch/plaque (T1/T2) disease — most early-stage patients are B0, and there is no clear survival difference between B0 and B1. Some guidelines therefore consider flow cytometry optional in T1/T2
It is essential in T3, T4 and any erythrodermic patient.
Putting it all toghether:
The four TNMB classifications combine to give a clinical stage from IA to IVB.
MF / Sézary Syndrome — Overall Stage Grouping
ISCL/EORTC TNMB stage groupings with defining clinical features.
| Stage | T | N | M | B | Defining feature |
|---|---|---|---|---|---|
| IA | 1 | 0 | 0 | 0–1 | Limited patches/plaques (<10% BSA) |
| IB | 2 | 0 | 0 | 0–1 | Generalised patches/plaques (≥10% BSA) |
| IIA | 1–2 | 1–2 | 0 | 0–1 | Patches/plaques + clinically abnormal nodes |
| IIB | 3 | 0–2 | 0 | 0–1 | One or more tumours |
| IIIA | 4 | 0–2 | 0 | 0 | Erythroderma, no/minimal blood involvement |
| IIIB | 4 | 0–2 | 0 | 1 | Erythroderma, low-level blood involvement |
| IVA1 | 1–4 | 0–2 | 0 | 2 | High-level blood involvement (Sézary range) |
| IVA2 | 1–4 | 3 | 0 | 0–2 | Effaced lymph nodes |
| IVB | 1–4 | 0–3 | 1 | 0–2 | Visceral involvement |
Early vs advanced:
IA, IB and IIA are early-stage and managed with skin-directed therapy
Note: IA or IB can have a small amount of blood involvement (B0,B1)
IIB onwards is advanced. The transition point is the appearance of a tumour, erythroderma, effaced nodes, B2 blood, or visceral disease.
Sézary syndrome: SS is, by definition, stage IVA1 or higher (T4 + B2). It is distinguished from erythrodermic MF (T4 with B0/B1) by the level of blood involvement.
MF / Sézary Syndrome — Investigations at Diagnosis & Staging
Recommended workup with stage-stratified indications.
| Investigation | When to do it |
|---|---|
| Skin exam, mSWAT, BSA | All patients |
| Lymph node palpation, organomegaly check | All patients |
| FBC, U&E, LFTs, LDH, immunoglobulins, HIV, HTLV-1/2 serology | All patients — HTLV-1 causes adult T-cell leukaemia/lymphoma (ATLL), which can mimic MF clinically and histologically; missing it changes prognosis and management |
| β2-microglobulin | Independent prognostic marker |
| Skin biopsy + TCR clonality | Diagnostic; repeat if non-diagnostic |
| Peripheral blood flow cytometry + TCR | T3, T4, erythroderma, suspected SS, palpable nodes; recommended in T2b; optional in T1/T2a |
| CT neck/chest/abdomen/pelvis | T3, T4, palpable adenopathy, B-symptoms, abnormal bloods; consider in T2b |
| FDG-PET-CT | Equivocal nodal disease, tumour-stage MF, to target the most avid node for biopsy |
| Excisional/core lymph node biopsy | Any clinically abnormal node >1.5 cm |
| Bone marrow biopsy | Unexplained haematological abnormality |
PROGNOSIS IN MF/SS
The prognosis in MF/SS is historically determined by TNMB stage, but stage alone does not capture all of the clinically important risk.
For instance early-stage MF usually has a favourable prognosis, particularly patch-only disease
However, some patients with early-stage disease have higher-risk features such as plaques, folliculotropic disease, abnormal nodes, older age or large-cell transformation. So two patients with the same stage can behave very differently
Also advanced-stage disease prognosis does not progress neatly by stage number. For example, stage IIB tumour-stage MF generally has a worse prognosis than stage IIIA erythrodermic MF without significant blood involvement
This is why prognosis is best thought of in two layers:
TNMB stage — how much skin, node, blood and visceral disease is present
Additional prognostic factors — clinical and pathological features that refine risk within each stage
Pearl:
Stage tells you where the disease is
Prognostic factors tell you how the disease is behaving
There are two main prognostic indices to help refine risk within stages:
CLIPi (Benton, EJC 2013) — retrospective analysis of 1502 MF/SS patients from the UK St John's cohort, identifying adverse prognostic factors for early- and advanced-stage disease
PROCLIPI (Scarisbrick, Blood 2025) — prospective international registry of >2000 patients across 46 expert centres, refining and validating the prognostic model for advanced disease
For early-stage disease (IA–IIA), the working model at time of writing (March 2026) remains CLIPi
PROCLIPI is still validating a prospective early-stage successor
Five adverse factors at diagnosis:
Male sex
Age >60
Plaques (T1b/T2b rather than patch only)
Folliculotropic disease
N1 or Nx
| Risk group | Factors | 10-year OS |
|---|---|---|
| Low | 0–1 | 90% |
| Intermediate | 2 | 76% |
| High | 3–5 | 49% |
In October 2025, PROCLIPI published its first dedicated prognostic index — for advanced disease — in Blood.
This is the most current, prospectively-validated model in the field, derived from 552 patients with stage IIB–IVB disease.
It identified four important adverse factors at diagnosis:
N3 nodal status (effaced nodes)
Age >60
Raised LDH
Large-cell transformation in skin
| Risk group | Factors | 5-year OS |
|---|---|---|
| Low | 0–1 | 63% |
| Intermediate | 2 | 45% |
| High | 3–4 | 18% |
The headline message is that risk group, rather than stage alone, predicts survival in advanced disease
High-risk patients (3–4 adverse factors) should be considered for more aggressive treatment escalation — including allogeneic stem cell transplant assessment — regardless of their precise stage
TREATMENT OF MF/SEZARY SYNDROME
The aim of this section is to give a practical framework for thinking about treatment in MF/SS, rather than an exhaustive list of every agent. The British Association of Dermatologists (BAD) and UK Cutaneous Lymphoma Group guideline (Gilson et al., BJD 2019) and the BMJ Best Practice review (regularly updated) are good reference points if you want to dig deeper
A few principles guide everything that follows:
MF/SS is, in most cases, a chronic, incurable disease. The aim of treatment is to control disease, manage symptoms (especially pruritus), and preserve quality of life — not to achieve cure.
Stage drives treatment. Early-stage disease (IA, IB, IIA) is managed with skin-directed therapy. Advanced disease (IIB and above) usually requires systemic therapy, often combined with skin-directed approaches.
Aggressive systemic chemotherapy in early-stage disease does not improve survival and adds toxicity. The pivotal Kaye et al. study (NEJM 1989) randomised early-stage patients to combination chemoradiotherapy versus sequential topical therapy and showed no survival benefit — this finding still underpins current practice.
Allogeneic stem cell transplant is the only potentially curative treatment but carries significant treatment-related mortality and is reserved for younger, fitter patients with advanced disease.
Cutaneous lymphoma analogy for patients:
The immune system is a bit like a factory with workers spread out across the body. The factory is the bone marrow — that's where the immune cells are made. Once they're made, they get sent out to where they're needed: some go to the lungs, some go to the gut, and a particular group is sent to patrol the skin.
What's happened in your case is that a small group of those skin-patrol cells has started copying itself when it shouldn't. That's why the problem is showing up as patches on the skin — the cells that have gone wrong are the ones that live there. The factory itself, and the rest of your immune system, is working normally.
Because this is happening in the skin and not in the factory, it tends to stay a skin problem, often for many years. We don't usually have a cure for it, but the aim is to keep it under control — and you'll notice that many of the treatments we use are the same ones we use for eczema and psoriasis: creams, light therapy, sometimes tablets. That's not because we're underestimating it; it's because those are the treatments that work.
Our aim is to:
Improve symptoms, cosmetic appearances and quality of life
Reduce the incidence of recurrence
What we worry about is if the misbehaving cells start to leave the skin and travel back through the body — into the lymph nodes, or the blood, or further. That's why we keep a close eye on your nodes and your bloods at every visit.
SKIN DIRECTED THERAPY
Skin-directed therapy (SDT) is the mainstay of early-stage disease and remains an important adjunct at all stages, particularly for symptom control.
Skin-directed therapies
Topical agents
Phototherapy
Radiation therapy
TOPICAL AGENTS
Emollients and “expectant therapy”
In very early, asymptomatic disease, an "expectant policy" with emollients alone is reasonable — particularly in older patients, those with minimal cosmetic concern, and those where treatment side effects may outweigh benefit.
Reducing scale and dryness will reduce pruritus.
Topical corticosteroids
Usually first-line active treatment.
Potent or very potent preparations (e.g. clobetasol propionate) for ≈ 6 weeks.
Reported response rates in case series are high (around 80–90% partial response), but durability is poor and recurrence on cessation is common.
Maintenance use is often needed.
Zackheim et al. (Arch Dermatol 1998) reported a complete response in 63% of T1 patients and 25% of T2 patients at a median of 9 months' follow-up.
Topical tacrolimus 0.1%
Has limited supporting data in the literature but is clinically useful for face and flexural patch disease where steroid use is limited.
Topical mechlormethamine (nitrogen mustard)
A DNA alkylating agent which was historically compounded as an ointment
Now there is a 0.02% gel formulation (Valchlor / Ledaga) which is licensed by the FDA and EMA. Lessin et al. (JAMA Dermatol 2013) showed response rates of around 58% in stage IA–IIA disease
Side effects: burning, irritation, contact dermatitis (in around 30%); managed with concomitant topical steroids, dose reduction, or reduced frequency.
Small percentage in original studies developed NMSC but further long-term studies have not shown a clear increase in non-melanoma skin cancer with the gel formulation
Carmustine (BCNU) is an alterantive topical alkylating agent
Topical bexarotene 1% gel
FDA-approved but not licensed in Europe (as far as I am aware)
Overall response rate 63% and clinical complete response rate 21% in early stage CTCL (Brenean D et al. Arch Dermatol. 2002 March)
Side effects: Mild to moderate irritation at application site that often improves with use (like other topical retinoids)
Other topicals (limited evidence)
Topical imiquimod — small case series only
PHOTOTHERAPY
Narrowband UVB (TL-01):
Effective for patch disease but limited dermal penetration means it is less effective for plaques.
One older study reported a complete response in 83% with median remission of 22 months (Ramsay et al., Arch Dermatol 1992).
The hypopigmented variant in younger patients tends to be particularly responsive
Relapse after stopping is common — sometimes within weeks — and patients should be counselled about this
PUVA:
Better penetration, so preferred for plaque-stage disease.
One of the most effective treatments for early disease, with response rates approaching 100% in some series.
More durable response than NBUVB, though formal head-to-head data are limited.
Can be combined with interferon-α or retinoids to improve response and reduce cumulative UV exposure.
Should cap at around 200 sessions lifetime to limit non-melanoma skin cancer risk
OTHER MODALITIES:
UVA-1 — small series, effective, but not widely available
Excimer laser (308 nm) — useful for localised disease
Clinical pearl:
Phototherapy is less effective in folliculotropic and syringotropic disease — the infiltrate sits too deep in the adnexa for UV light to reach
RADIOTHERAPY
MF is one of the most radiosensitive tumours we treat. Radiotherapy can be used at every stage of disease
Localised superficial radiotherapy:
It can be used for isolated lesions and palliation of bulky plaques or tumours
Relatively low doses are needed, eg:
Patches and plaques: 8 Gy in 2 fractions
Tumours: 12 Gy in 3 fractions.
"Curative intent" doses (higher fractionation) can be considered for solitary lesions of pagetoid reticulosis or unilesional MF
More complex techniques (e.g. tomotherapy, HDR brachytherapy) are used for awkward anatomy — for instance if the lesion is wrapping around limbs, digits, or the face
Pearl — the abscopal effect: Occasionally, radiotherapy to one site is followed by improvement in untreated lesions elsewhere. This appears to be an immune-mediated phenomenon and is an active area of clinical trial interest
Total Skin Electron Beam Therapy (TSEBT)
Is a radiation treatment involving electrons that treats the entire skin surface
95% of the radiation only penetrates into the first 2cm of the body to spare vital organs from radiation
It can be used for widespread plaque or tumour disease refractory to other SDT
A low-dose regiment TSEB (12 Gy in 8 fractions over 2 weeks) is now generally preferred over a previous higher dose (Morris et al., IJROBP 2017):
Why low-dose is preferred:
Shorter, less toxic
Allows future re-treatment with TSEB if needed.
Can be combined with systemic agents.
Practical considerations: patients need to be able to stand for 30–40 minutes and lift their shoulders. Lead shielding for testes, eyelids, and other sensitive sites
SYSTEMIC TREATMENT
Systemic therapy is generally indicated for:
Topicals and phototherapy are insufficiently effective
Relapse is rapid after phototherapy
Symptoms outweight the side-effect burden of escalation
Disease is widespread or located at particularly troublesome sites
Advanced-stage disease (IIB onwards).
A key counselling point: there is no robust evidence that systemic therapy in early-stage disease prevents progression or improves survival.
This is important to discuss with patients before escalation.
Choice of agent is guided by stage, comorbidities and patient preference
Systemic therapy
Established systemic agents
Newer targeted agents
Conventional chemotherapy
BEXAROTENE
Bexarotene (Targretin) is a synthetic rexinoid that selectively activates the retinoid X receptor (RXR) hence rexinoid
Licensed indication: advanced MF refractory to at least one systemic therapy. But it is often used off-licence in refractory early-stage disease.
Efficacy
Phase II/III data: Early-stage disease: ORR 54% (n=58); Subsequent retrospective series report response rates up to 83%
Response is dose-dependent, but dosing is limited by side effects.
Dosing
150-300mg/m2/day - typically 225-300mg/day starting dose and escalated 2-weekly as tolrated. Maximum response may take 6 months
Maintenance dose is typically 525-600mg/day
Adverse effects:
Hyperlipidaemia (≈ 83%) — triglycerides can climb to 10–12 mmol/L; Often may pretreat with fenofibrate prior to commencing it. May need to add other lipid lowering drugs if poorly controlled (eg statin, ezetimibe).
Central hypothyroidism (≈ 74%) — bexarotene suppresses TSH centrally. Pre-treat with levothyroxine (eg 25–50 mcg od) on day 1 of bexarotene. Monitor free T3/T4 and TSH together and adjust levothyroxine as required.
Neutropenia (≈ 47%).
Teratogenic — strict contraception required.
CYP3A4 interactions — including many lipid-lowering agents and azole antifungals.
Others - fatigue, dry skin/lips, headaches.
Baseline workup:
FBC, U&E, LFTs, lipid profile, CK, amylase, TFTs (TSH and T4), glucose.
Monitoring:
Two-weekly fasting bloods during dose escalations.
Interferon-α
Is a biological agent licensed for treatment of MF (can counsel patients this is not chemotherapy)
MoA is not not fully understood but it essentially wakes up the immune system and tips it back towards an anti-tumour response.
In MF, the malignant T cells are CD4+ helper cells with a Th2 profile — they produce cytokines that suppress the body's own anti-tumour immunity. Interferon-α counteracts this in three main ways:
Activates CD8+ cytotoxic T cells and NK cells — the cells that actually kill malignant lymphocytes
Suppresses the Th2 cytokine environment that the tumour cells thrive in, shifting the balance back towards a Th1 (anti-tumour) response
Increases visibility of the malignant cells to the immune system by upregulating MHC class I expression
Tend to use pegylated Interferon-α (subcut once weekly injection)
ORR ≈ 50% (up to 88%) in early-stage disease across multiple small non-randomised studies.
There are no formal MF/SS data for pegylated IFN, but efficacy is extrapolated from the non-pegylated formulations.
Adverse effects:
Flu-like symptoms after injection — pre-medicate with paracetamol 1 g 1 hour before
Depression — insidious onset over months; pre-existing depressive disorder is a contraindication
Thyroid dysfunction, hyperglycaemia
Neutropenia, transaminitis
Rare ocular changes — baseline eye test recommended
Caution with severe cardiac disease (HTN, arrhythmia, angina)
Methotrexate:
Single-agent low-dose oral methotrexate has reported response rates around 76% with 5-year survival around 71% (older series).
It is a useful systemic option in MF/SS, particularly when an oral, lower-intensity treatment is preferred.
Higher response rates (based on retrospective disease) appear to be seen in erythrodermic MF rather than tumour-stage
Practical advantage: easier to monitor and prescribe than bexarotene or interferon
EXTRACORPOREAL PHOTOPHORESIS (ECP)
A "systemic form of PUVA".
Use in specialist centres.
It is primarily used in erythrodermic MF and Sézary syndrome, particularly where there is significant blood involvement.
It has a favourable tolerability profile and is often combined with interferon-α or bexarotene.
Responses may take several months, and it is generally less effective for bulky tumour-stage or transformed disease.
How it works:
Mononuclear cells are isolated by leukapheresis
They are then exposed to 8-methoxypsoralen and then UVA, and reinfused back into the patient
First-line for erythrodermic MF and Sézary syndrome
Can be used in combination with other treatments (eg IFN-α, bexarotene)
Typically given over 2 days every 2–4 weeks.
The UVA is directly toxic to irradiated cells and reinfused cells also appear to stimulate selective immune response against the malignant cells
Favourable factors for ECP response:
Short disease duration (preferably < 2 years)
Absence of bulky lymphadenopathy or major visceral involvement
White cell count < 20,000/μL
A discrete (not overwhelming) burden of Sézary cells (10–20% of mononuclear cells)
No prior intensive chemotherapy
Histone deacetylase inhibitors (eg vorinostat and romidepsin)
HDAC inhibitors are systemic anti-cancer treatments, but they are better described as targeted/epigenetic therapies rather than conventional chemotherapy.
They work by altering how lymphoma cells switch genes on and off by inducing histone and protein hyperacetylation
This can lead to cell cycle arrest and apoptosis.
Useful particularly for pruritus control as well as skin disease.
In the MAVORIC trial (see below), mogamulizumab was significantly superior to vorinostat for both PFS and ORR and vorinostat has been somewhat displaced as a standard comparator.
Brentuximab vedotin
Antibody–drug conjugate that targets CD30
ALCANZA trial (Prince et al. Lancet 2017): Phase 3 RCT, 131 patients with CD30+ MF (stage IB–IV) or pcALCL, randomised to brentuximab vs physician's choice (methotrexate or bexarotene):
Objective global response > 4 months: 56% vs 13%
Complete response: 16% vs 2%
Progression-free survival: 16.7 vs 3.5 months
Important practical points:
Access in Ireland and the UK requires documented CD30 positivity on biopsy.
CD30 expression in MF is variable across biopsy sites; multiple biopsies may be needed before CD30 positivity is obtained.
Initial response may be a flare; median time to response ≈ 12 weeks.
Useful as a bridge to alloHCT in advanced disease — small series show high response rates pre-transplant.
Adverse effects:
Peripheral neuropathy — reported in up to 45%, can be severe (motor and sensory). Dose reduce or stop early at the first signs.
Infusion reactions
Tumour lysis (reflecting how active the drug can be)
Drug rash, fatigue
Mogamulizumab
A first-in-class defucosylated humanised anti-CCR4 monoclonal antibody.
CCR4 is highly expressed on malignant T cells in CTCL, particularly in blood-involved disease.
Defucosylation (removal of a sugar called fucose during manufacturing of the antibody) enhances antibody-dependent cellular cytotoxicity (ADCC) up to 100-fold compared with conventional mAbs.
MAVORIC trial — phase 3 RCT, mogamulizumab vs vorinostat in previously treated CTCL:
PFS: 7.7 vs 3.1 months (HR 0.53; p < 0.0001)
ORR: 28% vs 5% (p < 0.0001)
Significantly improved patient-reported quality of life (Skindex-29)
Particularly effective in Sézary syndrome and erythrodermic disease with blood involvement — response rates and durations are higher in blood/erythrodermic disease than in skin-only disease.
Adverse effects: Infusion reactions and drug eruptions (which can mimic disease progression — important to recognise).
Alemtuzumab
Anti-CD52 monoclonal antibody.
ORR ≈ 55% (32% CR) in advanced disease in older series. Reserved for refractory disease, generally after newer agents have failed.
Significant infection risk (CMV reactivation, opportunistic infection) — careful monitoring required.
CONVENTIONAL CHEMOTHERAPY
Chemotherapy is generally reserved for advanced, rapidly progressive, transformed, or refractory MF/SS after better-tolerated systemic options have been considered.
Combination chemotherapy, such as CHOP or EPOCH, can produce high initial response rates, but responses are usually short-lived, toxicity is significant, and there is no clear survival benefit.
When chemotherapy is needed, single-agent treatment is usually preferred over combination regimens. Options include gemcitabine, doxorubicin and purine analogues such as pentostatin, cladribine or fludarabine.
In general, chemotherapy in MF/SS is palliative rather than curative.
CD 30+VE LYMPHOPROLIFERATIVE DISORDERS
CD30 is a protein made by activated T and B lymphocytes
It is present in CD30 +ve lymphoproliferative disorders, some cases of MF and Hodgkin’s lymphoma
LYMPHOMATOID PAPULOSIS
Commonest ‘CTCL’ in children
Technically not a lymphoma but confers and increased risk for cutaneous lymphoma
Get recurrent papules and/or nodules that frequently cruts or ulcerate and then heal often with atrophic hypopigmented scarring
They are often asymptomatic, rapidly growing skin lesions that involute over about 6-10 weeks
Often can be mistaken for acne/insect bites
Predominantly on trunk and limbs but can present anywhere including palms and soles
Can get lesions at different stages of evolution
No associated systemic symptoms
Ddx: insect bites, acne, PLEVA, molluscum
May develop an associated systemic lymphoma in 2-10% of patients so it very important that they are kept under surveillance
HISTOPATHOLOGY
Atypical lymphocytes in dermis with plemorphic and hyperchromic nuclei
May see mixed infiltrate
CD30+ve in majority of cases
Depends on when biopsy taken as biopsy at any one time only shows a snapshot in time of an evolving process
4 histological subtypes have been identified (A-D)
Most common subtype is type A in which you see large, atypical CD30+ve lymphocytes in a wedge-shaped distribution throughout the dermis and ixed with inflammatory cells
ANAPLASTIC LARGE CELL LYMPHOMA
Part of the spectrum of CD30+ve CTCL
Typically get a solitary tumour which may increase in size and ulcerate
HISTOPATHOLOGY
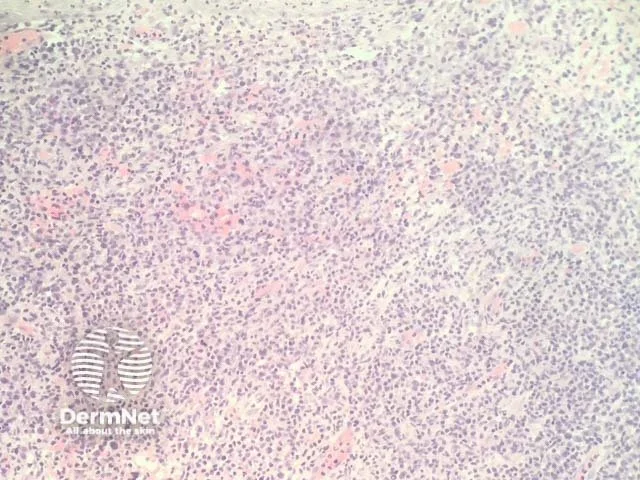
Histological features of PC-ALCL can resemble those seen in anaggressive systemic lymphoma but the disease doesn’t follow an aggressive course
Confluent sheets of large, atypical lymphocytes infiltrating dermis and subcutis
Don’t tend to see epidermotropism
Mitotic figures abundant
Other ddx:
Can share histological features with Lymphomatoid papulosis
MF can show large cell transformation and can be CD30+ve (if have multiple patches and plaques consider MF)
IMMUNOHISTOCHEMISTRY
CD30+ve cellswhich can be CD4+ or CD8+ve
(may also express CD3 and CD5)
It is essential to distinguish primary cuataneous anaplastic large cell lymphoma (PC-ALCL) from systemic ALCL
The absence of staining with anaplastic lymphoma kinase (ALK) favours the primary cutaneous variant
(Think the absence of staining means it is absent elsewhere)
ALK staining is detected in > 90% of systemic ALCLs and results from a translocation involving the ALK gene causing the expression of the ALK protein
(Translocation is detectable with cytogenetic tests)
TREATMENT
Lesions can be excised or treated with radiotherapy
Spontaneous regression may occur
Multiple cutaneous lesions or nodal spread may require systemic treatment
Brentuximab vedoin (Adcetris):
Granted FDA approval November 2017 for treatment of adult patients with:
Primary cutaneous anaplastic large cell lymphoma or CD30 +MF who have received prior systemic therapy
ALCANZA trial:
Phase 3 trial
Looked at patients with CD 30+ cutaneous T-cell lymphomas (either CD30+ MF or pc-ALCL)
A significant improvmeent in objective global response was seen with brentuximab versus physician’s choice of mehtotrexate or bexarotene
Objective global resposne: 56% vs 13%
Complete response: 16% vs 2%
Progression free survival: 16.7 months vs 3.5 months
PROGNOSIS
Generally quite good
5-year survival rates above 90%
Topics to do:
Rarer sub-types of T cell lymphoma
Subcutaneous panniculitis like T cell lymphoma:
Commonest cause of panniculitis is E nodosum
What are the things that make panniculitis atypical:
Persistance vs recurrent episodes with complete clearing in between
Tend to be on lower extremities also
On pathology tends to be a lobular panniculitis (similar to lupus panniculitis)
Will have atypical cells on H&E
Can be difficult to distinguish between lupus panniculitis and subcutaneous panniculitis like T cell lymphoma
Some consider them to be two entities in a spectrum
About 20% of patients with subcutaneous panniculitis T cell lymphoma have positive ANA
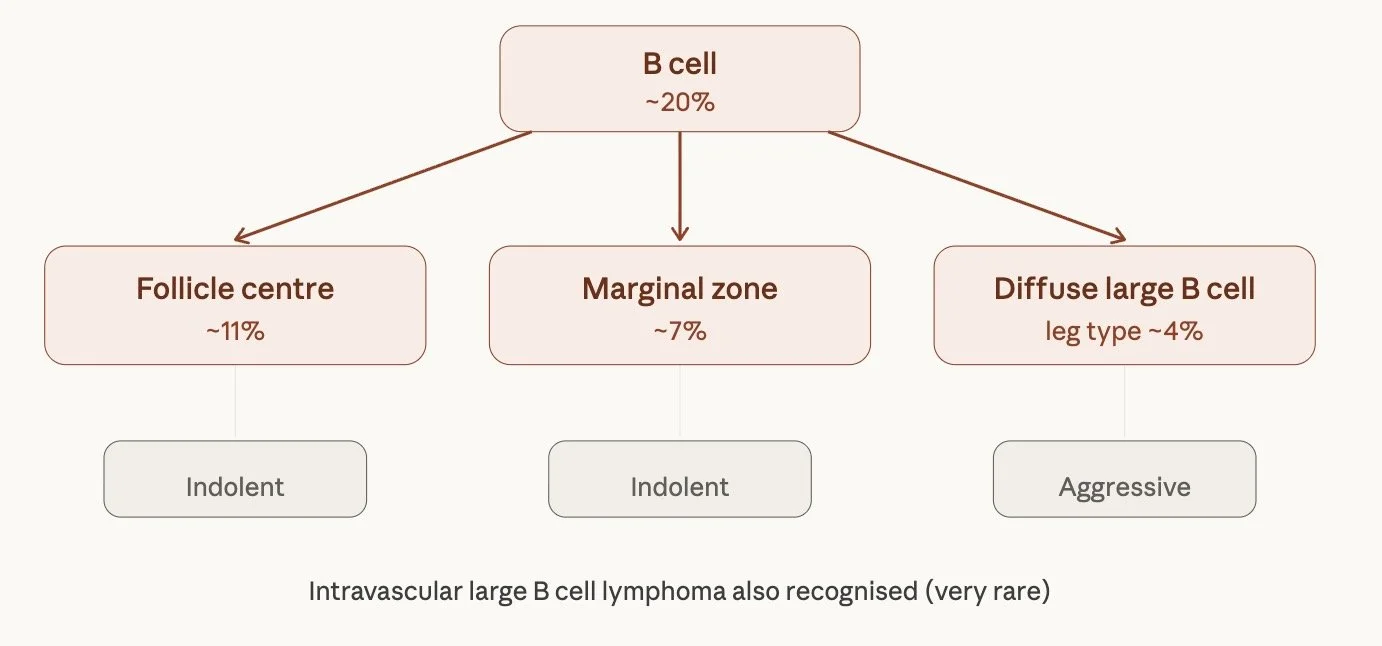
B CELL LYMPHOMA
Less than 1/3 of primary cutaneous lymphomas
Main types:
Primary cutaneous:
Marginal zone B-cell lymphoma
Follicle centred B-cell lymphoma
Diffuse large B-cell lymphoma
Don’t tend to have the epidermal changes with a scaly rash with ulceration that T cell lymphomas tend to have have
Patients often present with plum coloured/reddish brown dome shaped nodules
In most cases they are asymptomatic
Can be isolated lesions or a few scattered lesions
Low grade B cell lymphomas tend to be on the head or neck or upper trunk
The more aggressive B cell lymphoma (large anaplastic B cell lymphoma) tend to be on lower extremities
Site localization can be an important distinguishing feature
Borrelia burgoderfi has been implicated in the pathogenesis of all types of CBCL in endemic areas in Europe
Serology is indicated in all patients
PRIMARY CUTANEOUS FOLLICLE CENTER LYMPHOMA
55% of all CBCL
Firm plum coloured dermal lesions over trunk or head
Salp lesions may be associated with alopecia
Multiple lesions may occur and relapse is frequent
Systemic spread is rare
It is very important to be aware that secondary cutaneous invovlement with systemic follicle centre lymphoma can occur so full staging investigations are essential to ensure you are dealing with a primary cutaneous lymphoma
HISTOPATHOLOGY
Diffuse, folliclular or mixed pattern of large centrocytes and centroblasts in the dermis without epidermotropism
IMMUNOHISTOCHEMISTRY
CD19, CD20, CD22, CD79, BCL-6 positive
(CD10 may be positive in some lesions)
In a systemic follicular lymphoma the t(14;18) translocation often found but typically absent in primary cutaneous FCL
TREATMENT
Usually skin directed therapy
If have multiple lesions could consider rituximab or low dose chemotherapy (chlorambucil)
Clonal IgH gene rearrangements can aid diagnosis
B cell lymphoma pathology:
Multiple other markers used to look at to see what type of cell it is (eg CD-20, bcl-2, bcl-6)
Borrelia burgoderfi serology (reported in associated with PCMZL
B cells have immunoglobulins (antibodies) instead of a T cell receptor
Can look for clones that have identical immunoglobulin heavy chains
Clonal IgH gene rearrangements can aid diagnosis in B cell lymphoma
Management:
Low grade lymhomas:
Prognosis tends to be very good
Do blood work and imaging
If it is confirmed that this is a primary cutaneous low grade B cell lymphoma
Options could include:
Monitor
Intralesional steroids
Excise if one lesion
Radiation if larger lesion
Aggresive B cell lymphoma:
Refer urgently to haematology for management
Often has poor prognosis
Staging cutaneous lymphomas other than MF/SS:
In 2007, a staging system was developed for all ohter cuatnous lymphomas that describes the extent of the lymphoma (using a TNM classification) but it does not give any useful information about prognosis
I won’t go into much more detail about that here
Issues:
Many different type of cutaneous lymphomas
Most are rare
Not useful in clinical practice
Reason for classification:
Current stating system helps classify patients
Over time may be able to better define prognosis for different types of cutnaeous lymphoma
Currently it is easier to define prognosis based on growth pattern of the lymphom
Indolent:
Grows slowly
Treatment may not be necessary unless causing symptoms
Often not curable
Aggressive:
Grow rapidly
Treatment necessary for survival
Often curable
Parapsoriasis:
Pseudolymphoma:
Term represents several clinical entitiies that probably have multiple aetiologies
Lymphocytoma
Spiegler-Fendt sarcoid
Lymphadnosis benigna cutis
Jessner’s benign lymphocytic infiltrate
In most cases aetiology is unknown (some cases may be related to chronic arthropod bites)
Usually present as indloent single or grouped red/purple nodules or plaques on the head, neck and upper trunk
Infiltrate may show B or T cell predominance
Some cases can be difficult to differentiate from lymphoma and patient should be follwed up prior to definitive diangosis being made
Leukaemia cutis:
CLL in adults is the most common cause of specific leukemic skin lesions in adults
They are usually multiple and may present wih papules, nodules, plaques, erythema and rarely bullae
The neoplastic proliferation of lymphocytes is usually B cell in origin